Glioblastoma (GBM) is the most common and malignant primary brain tumor in adults, with a median patient survival of less than 20 months and extremely poor prognosis. The current standard of care includes surgical resection, temozolomide chemotherapy, and radiotherapy. Among these, radiotherapy represents the most effective first-line treatment to prolong patient survival. However, nearly all patients eventually succumb to tumor recurrence due to radiotherapy resistance, which is closely associated with a subpopulation of glioblastoma stem cells (GSCs) characterized by self-renewal capacity and intrinsic radioresistance. Overcoming such radiotherapy resistance has become a critical challenge to improve patient outcomes.
Recently, in collaboration with New York University, Duke University, and other institutions, professor Zhang Ze-Yan from the School of Basic Medicine/ Institute of Biomedical Innovation, Nanchang University, published a research article in Nature Communications entitled “NANP targeting radiosensitizes glioblastoma through TNFR1 sialylation-driven mesenchymal shift”. This study reports that NANP, a key enzyme in the sialic acid biosynthetic pathway, acts as a potential target for radiosensitization by regulating sialylation and endocytosis of tumor necrosis factor receptor 1 (TNFR1), activating the NF-κB signaling pathway, and driving the mesenchymal (MES) phenotypic transition of GSCs.

Using patient-derived GSCs as models, the team first employed single-cell DNA barcode lineage-tracing technology and revealed that radiotherapy resistance, unlike targeted therapy resistance, does not involve significant enrichment of specific resistant subclones, suggesting that “global radiosensitization” represents a more feasible therapeutic strategy. Further genome-wide CRISPR screening identified NANP, a key enzyme in the sialic acid synthetic pathway, as a top uncharacterized radiosensitizing target, whose high expression correlates with tumor radioresistance. Functional assays demonstrated that silencing NANP markedly enhances radiation-induced DNA damage, G2/M cell cycle arrest, and apoptosis, thereby improving the radiosensitivity of GBM. Mechanistically, NANP modulates TNFR1 sialylation and endocytosis to activate NF-κB signaling, drives the MES phenotypic shift in GSCs, and ultimately promotes radiotherapy resistance in GBM.
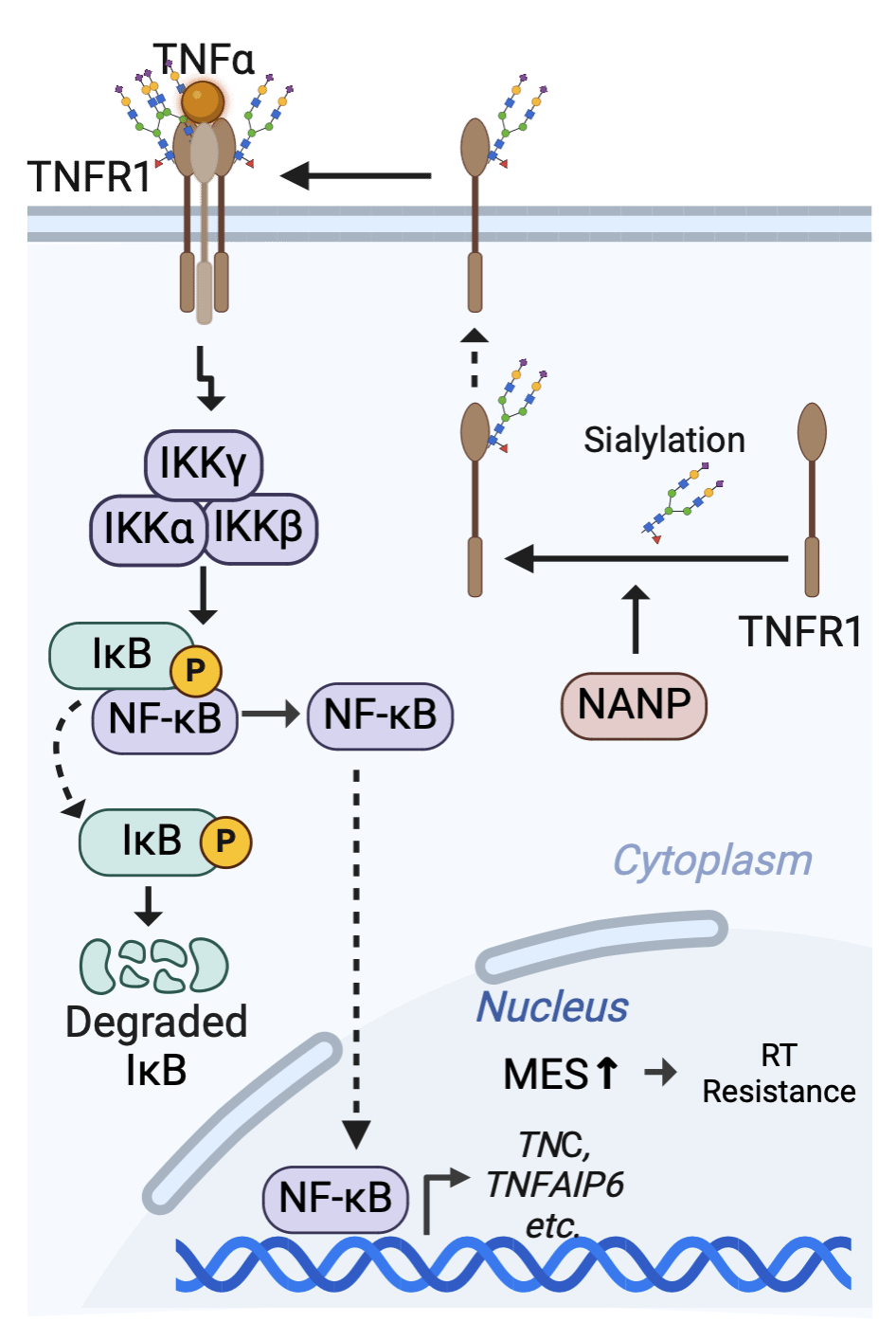
This study elucidates the mechanism underlying radiotherapy resistance in glioblastoma and provides a critical theoretical basis and potential target for the development of novel radiosensitizing agents.
Prof. ZHANG Ze-Yan (School of Basic Medicine, Nanchang University) and Dr. Yingwen Ding are co-first authors of this paper. Prof. Erik P. Sulman, Interim Director of the Duke Cancer Institute and Chair of the Department of Radiation Oncology, is the corresponding author. Nanchang University is the first affiliation.
Original link: https://www.nature.com/articles/s41467-026-70853-x
